Gene Therapy for ARVC
The role of genetics
To understand gene therapy as a potential treatment for ARVC, it helps to know some basic information about genetics.
Cells are the basic building blocks of all living things.1 They contain many different parts, including a person’s genetic information.
Scroll through the illustrations below to learn more about the cell structures.
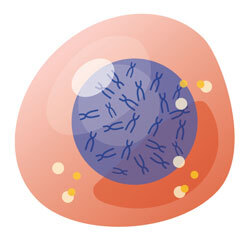
Cells
The human body is made up of trillions of cells. Cells contain many parts including chromosomes.

Chromosomes
Chromosomes are long strands of DNA.2

DNA
Within DNA are genes.

Gene (section of DNA)
Genes contain the instructions to make proteins.

Proteins
Proteins work together to tell your body how to grow, develop, and function.3,4 For example, proteins are involved with making your organs—including your liver, lungs, and heart—work.4
How changes in a gene can cause genetic conditions
Mutations (also known as variations or changes) in a gene can affect that gene’s ability to make a given protein the way it should or can affect whether that protein is made at all. In some cases, too much or too little of a necessary protein can result in a disease or condition.
In genetic ARVC, mutations can prevent heart muscle cells from making enough or any protein needed to maintain their structural connections and communication channels. Over time, these cells become weaker and are replaced with fatty and scar tissue, and the electrical pulses can become unstable and cause arrhythmias. The most common cause of ARVC is due to mutations in the PKP2 gene.5 For more information about the genetic causes of ARVC, click here.
What is gene therapy?
Gene therapy is a way of treating or preventing diseases or medical conditions caused by genetic mutations. Gene therapy is intended to deliver a working gene into a cell to help the cell build the necessary protein and restore the expected function.6
There are several types of gene therapy, and each one uses a different approach to address disease causing mutations.7
- Gene replacement therapy adds a working to supplement or replace the function of a non-working gene to address the underlying cause of a condition. It is also called gene addition because this approach adds a working gene into target cells.8
- Think of this as delivering a new set of “genetic instructions” into a person’s cells. This can be done by using a capsid (also known a vector) as a vehicle to deliver a working gene into cells.
- Tenaya’s investigational treatment for ARVC is a gene replacement therapy called TN-401 designed to deliver a working PKP2 gene to specific muscle cells that make the heart contract and relax with each beat.
- Gene editing makes changes in the DNA to correct (or edit) faulty information caused by a mutation8
- Think of this as rewriting the “genetic instructions” within a person’s DNA. Gene editing may be used when a “misspelling” of a gene results in a genetic condition. The genetic misspelling (or mutation) is corrected to address the root cause of the genetic condition.9
Gene editing is a different approach to addressing the precise underlying cause of disease, but researchers are still learning about its potential in genetic heart conditions.
Why are AAV capsids used in gene therapy?
- it is efficient at delivering new genes to cells, and
- there are different types of AAVs that can be tailored to target specific types of cells (such as heart muscle cells)
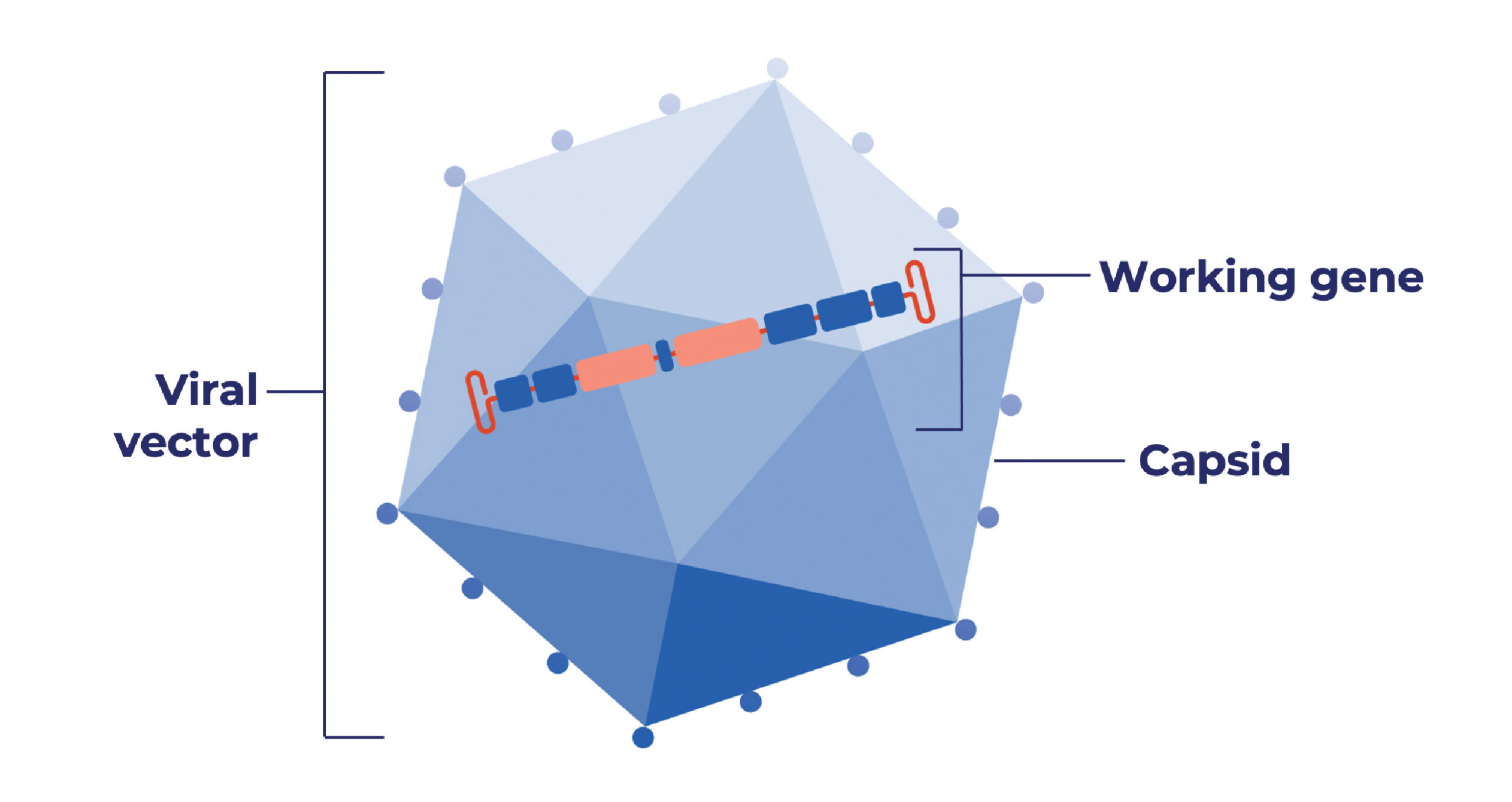
When used in gene therapy, AAV uses a capsid (also called a vector) as a vehicle to deliver a working gene into cells. AAV gene therapy11:
- Removes all viral genes that could cause disease or illness, leaving the capsid
- Adds a working gene to specific cells
- Does not change or edit a person’s own DNA
- Does not make changes to a person’s sperm or egg cells (and therefore, the gene therapy cannot be passed from a parent to a child)
- Will not prevent or lower a person’s risk of passing a genetic condition to their children
As of November 2024,
The U.S. FDA has approved six AAV gene therapies17:
- A treatment for people with inherited retinal disease (IRD)
- A treatment for children less than two years old with spinal muscular atrophy (SMA)
- A treatment for adults with hemophilia B
- A treatment for children aged four to five with Duchenne muscular dystrophy (DMD)
- A treatment for adults with severe hemophilia A
- A treatment for adults and children with aromatic L-amino acid decarboxylase (AADC) deficiency
The European Medicines Agency has authorized approval in the European Union for four AAV gene therapies:
- A treatment for adults with severe hemophilia A18
- A treatment for adults and children aged 18 months and older with severe aromatic L-amino acid decarboxylase (AADC) deficiency19
- A treatment for children with spinal muscular atrophy (SMA)20
- A treatment for an inherited retinal (eye) disease (IRD) that causes blindness21
About Tenaya’s TN-401 gene therapy for ARVC due to PKP2 mutation
TN-401 is an investigational gene therapy designed to reach the specific muscle cells that make the heart contract and relax with each beat.
- TN-401 uses a specific type of AAV capsid called AAV9.
- The AAV9 capsid has been used to treat thousands of people around the world with gene therapy. The AAV9 capsid has been extensively studied in thousands more in clinical trials, including for heart disease.8,14
- The AAV9 capsid used in TN-401 is designed to deliver a working PKP2 gene to the muscle cells in the heart
- Once in these cells, the working PKP2 gene delivered by TN-401 may help make the protein needed to restore typical heart function
- TN-401 is given as a one-time intravenous (IV) infusion
How TN-401 Works
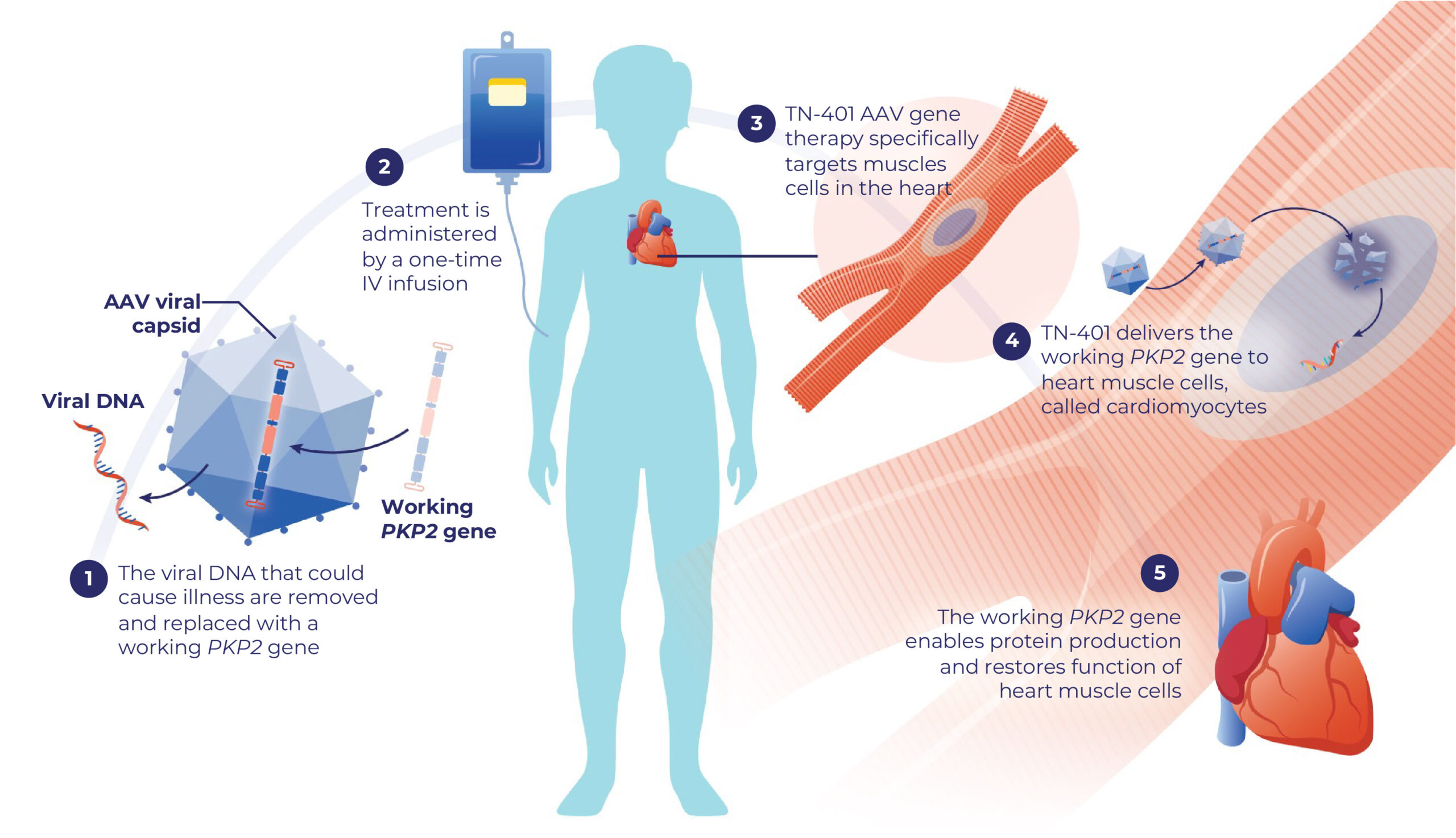
This brochure will help you understand how Tenaya’s gene therapy may help heart cells make protein.
Learn more about the clinical trial for TN-401 in people with ARVC due to PKP2 mutation.
From Gene to Protein
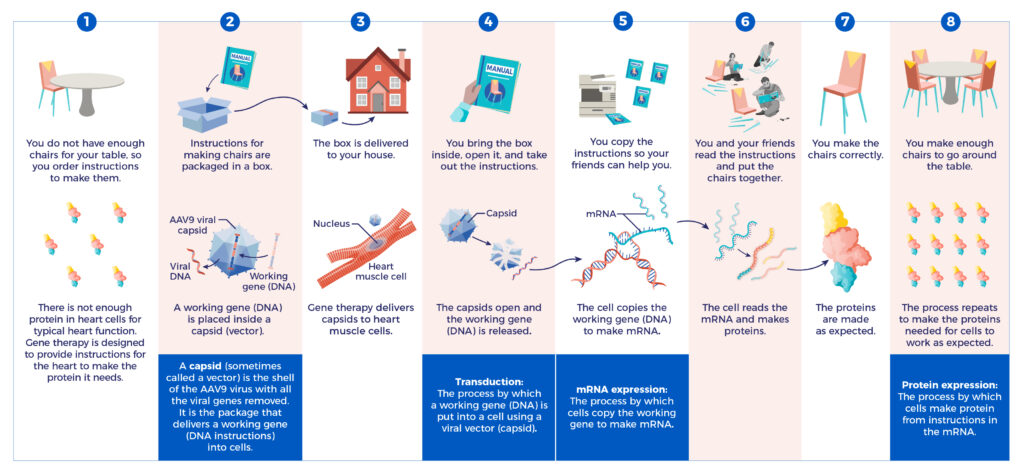
Tenaya’s gene therapies are investigational and have not been approved by the U.S. Food and Drug Administration (FDA) or any other country’s health authority or regulatory agency.
Expand for References
1. Studying cells. U.S. Department of Health and Human Services, National Institutes of Health, National Institute of General Medical Sciences; 2023.https://www.nigms.nih.gov/education/Pages/Glossary.aspx#letter-C . Accessed April 15, 2024. 2. Glossary. American Society of Gene + Cell Therapy; 2023. https://asgct.org/education/more-resources/glossary. Accessed April 15, 2024. 3. Gene. U.S. Department of Health and Human Services, National Institutes of Health, National Human Genome Research Institute; 2024. https://www.genome.gov/genetics-glossary/Gene. Accessed April 15, 2024. 4. Protein. U.S. Department of Health and Human Services, National Institutes of Health, National Human Genome Research Institute; 2024. https://www.genome.gov/genetics-glossary/Protein. Accessed April 15, 2024. 5. Jacob KA, Noorman M, Cox MG, et al. Geographical distribution of plakophilin-2 mutation prevalence in patients with arrhythmogenic cardiomyopathy. Neth Heart J. 2012;20(5):234-239. 6. Gene Therapy. U.S. Department of Health and Human Services, National Institutes of Health, National Human Genome Research Institute; 2024. https://www.genome.gov/genetics-glossary/Gene-Therapy. Accessed April 15, 2024. 7. Vectors 101. American Society of Gene + Cell Therapy. 2024. https://patienteducation.asgct.org/gene-therapy-101/vectors-101. Accessed April 15, 2024. 8. Gene Therapy Approaches. American Society of Gene & Cell Therapy; 2023. https://patienteducation.asgct.org/gene-therapy-101/gene-therapy-approaches. Accessed April 15, 2024. 9. Gene Editing. American Society of Gene + Cell Therapy. 2021. https://patienteducation.asgct.org/gene-therapy-101/gene-editing. Accessed April 15, 2024. 10. Li C, Samulski RJ. Engineering adeno-associated virus vectors for gene therapy. Nat Rev Genet. 2020;21(4):255-272. https://doi.org/10.1038/s41576-019-0205-4. 11. Naso MF, Tomkowicz B, Perry WL 3rd, Strohl WR. Adeno-associated virus (AAV) as a vector for gene therapy. BioDrugs. 2017;31(4):317-334. https://doi.org/10.1007/s40259-017-0234-5. 12. Kuzmin, DA, Shutova MV, Johnston NR, et al. The clinical landscape for AAV gene therapies. Nat Rev Drug Discov. 2021;20(3):173-174. https://doi.org/10.1038/d41573-021-00017-7. 13. Au HKE, Isalan M, Mielcarek M. Gene therapy advances: a meta-analysis of AAV usage in clinical settings. Front Med (Lausanne). 2022 ;8 :809118. https ://doi.org/10.3389/fmed.2021.809118. 14. George LA, Ragni MV, Rasko JEJ, et al. Long-term follow-up of the first in human intravascular delivery of AAV for gene transfer: AAV2-HFIX16 for severe hemophilia b. Mol Ther. 2020;28(9):2073-2082. https://doi.org/10.1016/j.ymthe.2020.06.001. 15. Novartis. Q3 2022 Results; 2022. Accessed March 29, 2023. 16. Keeler AM, Flotte TR. Recombinant adeno-associated virus gene therapy in light of Luxturna (and Zolgensma and Glybera): where are we, and how did we get here? Annu Rev Virol. 2019;6(1):601-621. https://doi.org/10.1146/annurev-virology-092818-015530. 17. Approved cellular and gene therapy products. U.S. Food and Drug Administration (FDA); 2024. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/approved-cellular-and-gene-therapy-products. Accessed April 15, 2024. 18. Roctavian. European Medicines Agency; 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/roctavian. Accessed April 15, 2024. 19. Upstaza. European Medicines Agency; 2024. https://www.ema.europa.eu/en/medicines/human/EPAR/upstaza. Accessed April 15, 2024. 20. Zolgensma. European Medicines Agency; 2024. https://www.ema.europa.eu/en/medicines/human/EPAR/zolgensma. Accessed April 15, 2024. 21. Luxturna. European Medicines Agency; 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/luxturna. Accessed April 15, 2024.